白血病と悪性リンパ腫
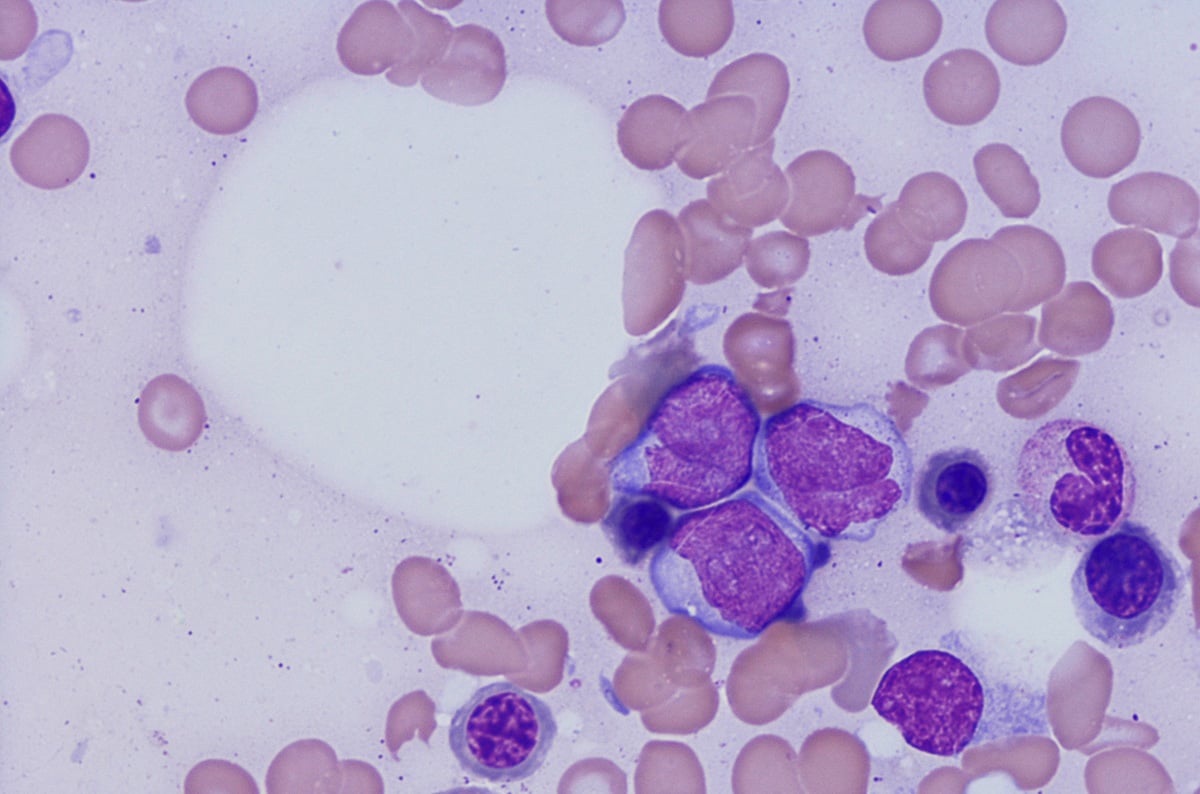
造血器腫瘍(血液がん)は免疫系細胞の疾患であり、腫瘍が最初に見つかった場所が血液中であれば白血病2、リンパ節ならホジキンリンパ腫や非ホジキンリンパ腫3、骨髄の場合は骨髄腫4と分類されます。

がんの罹患者数と死亡者数は世界中で急速に増加しており、その原因として人口の高齢化および増加、主要リスク因子を持つ人の割合や分布の変化が挙げられます1。血液がんは全てのがんの約6%を占め、2020年に新たに血液がんと診断された人は120万人、血液がんによる死亡者は70万人近くにのぼります1。血液がんが増加していることで、早期に的確な診断と特性解析を行い、少しでも早く最善の治療を開始することがますます重要になっています。
血液がんの種類
白血病は、血液細胞ががん化(腫瘍化)することで引き起こされます。このがん化は、造血幹細胞から血液細胞へ分化する造血の過程において、いつでも起こり得ます。このがん化が起こる時点は、白血病の種類によって異なるため5、どの種類の白血病であるかを正確に分類するには細胞系統を調べることが非常に重要です。白血病は、がん化した血液細胞の種類によって、リンパ腫または骨髄腫に分類され、疾患増悪率によって、急性または慢性に分けられます2。
リンパ腫はリンパ球ががん化することで起こります。表面マーカー、腫瘍構造、細胞形態、分化、遺伝子変異によって、多くのサブタイプに分類されます6 。最適な治療を選択するには、このサブタイプを早期に見極めることが重要です。

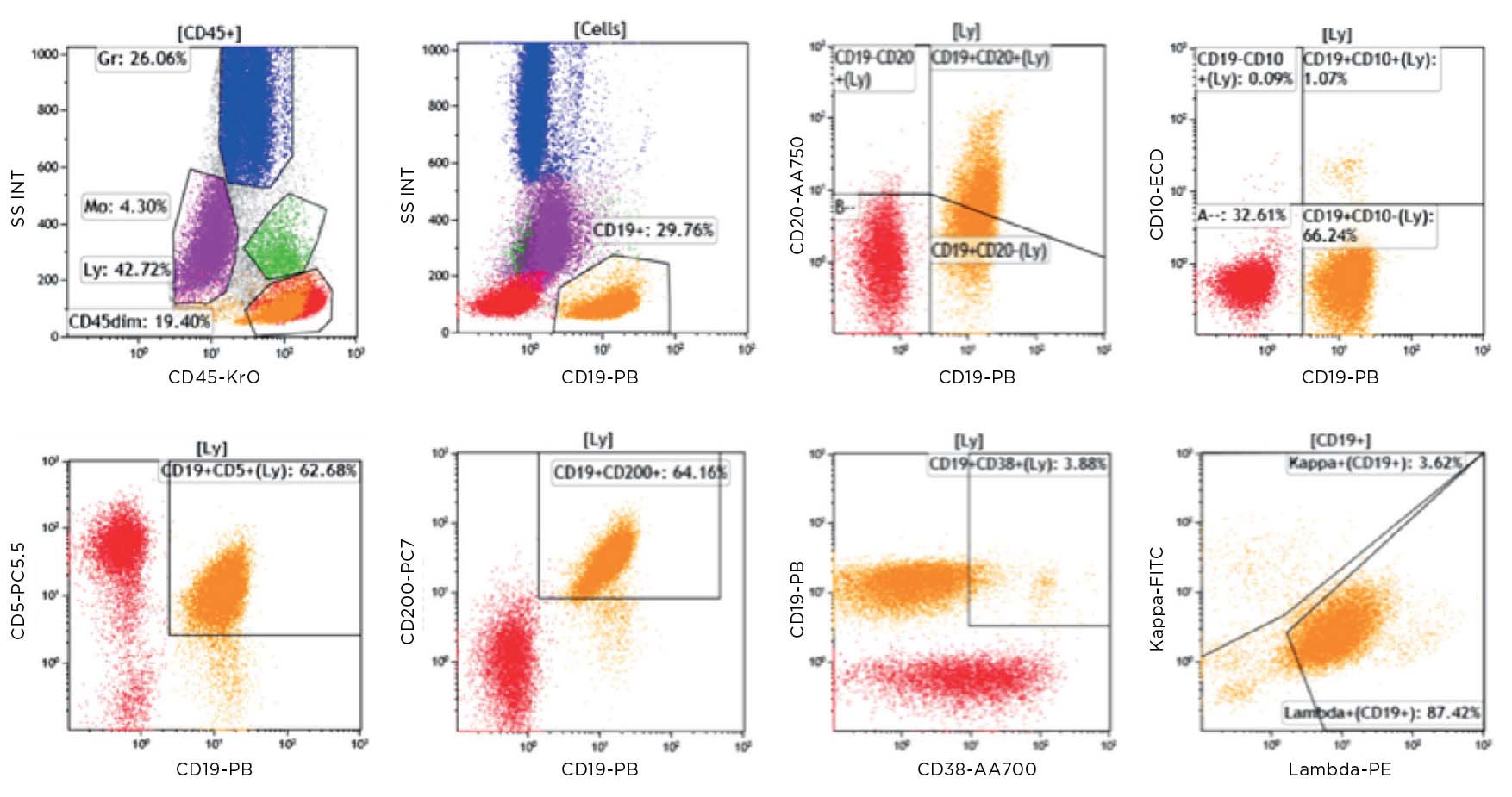
フローサイトメトリーによるイムノフェノタイピング(Flow cytometric immunophenotyping, FCI)は、造血器腫瘍の診断、分類、ステージ判定、モニタリングに有用なツールです。FCIでは抗体パネルで細胞のマーカーを検出し、腫瘍細胞とその系統を決定します。FCIは抗原発現量のわずかな違いを検出できるため、ごく少数しか存在しない異常細胞を見分けることが可能です。従来、血液がんの診断に用いられてきた免疫組織染色(IHC)法と比較した場合、フローサイトメトリーには以下のような利点があります。
- 細胞径と粒度から細胞群を同定
- 死細胞を識別して分析から除外
- 発現の弱い細胞表面抗原を検出
- マルチカラー解析で、複数の細胞内・表面抗原を同時に測定
- 迅速な測定結果
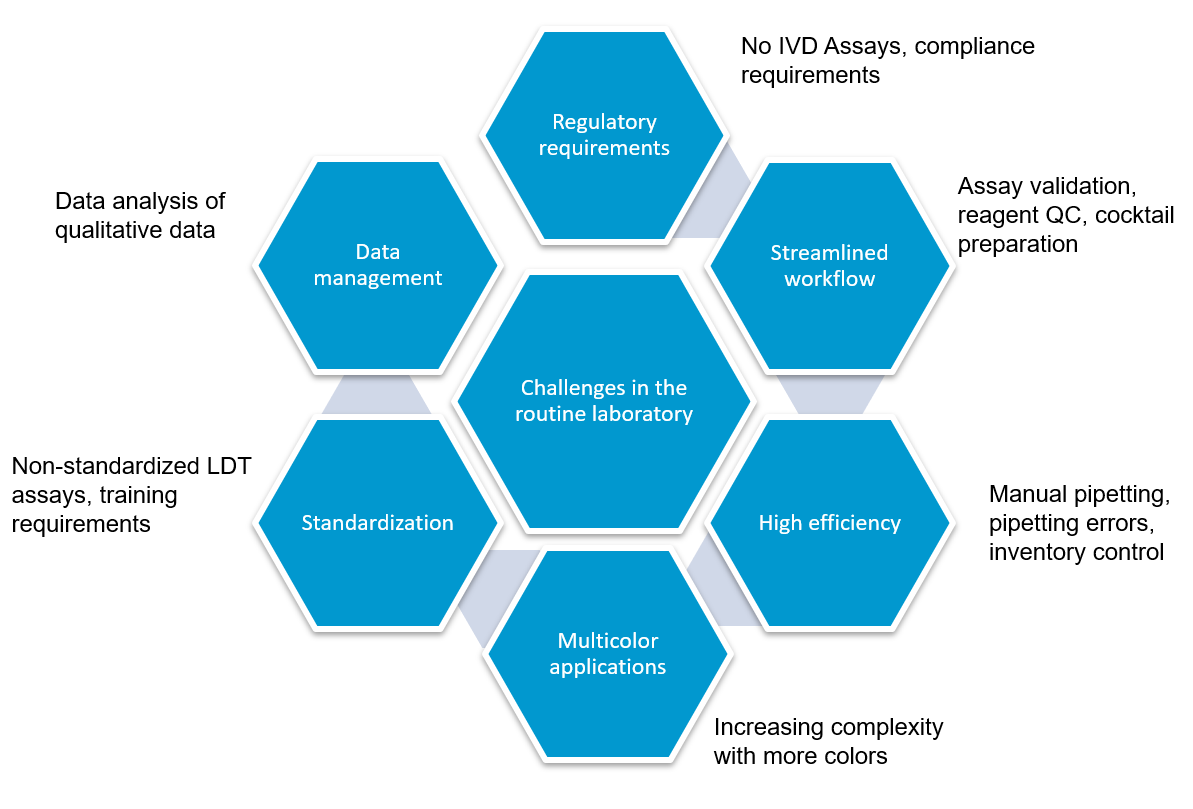
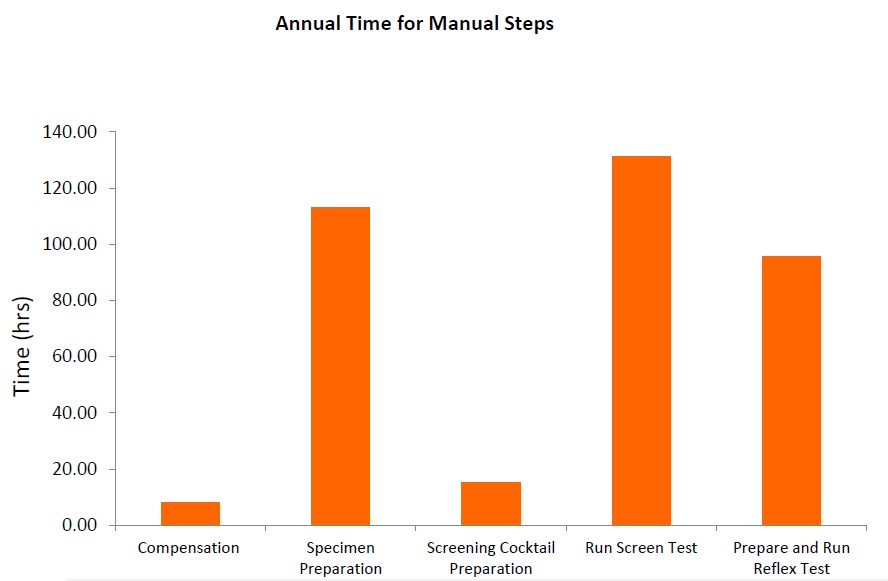
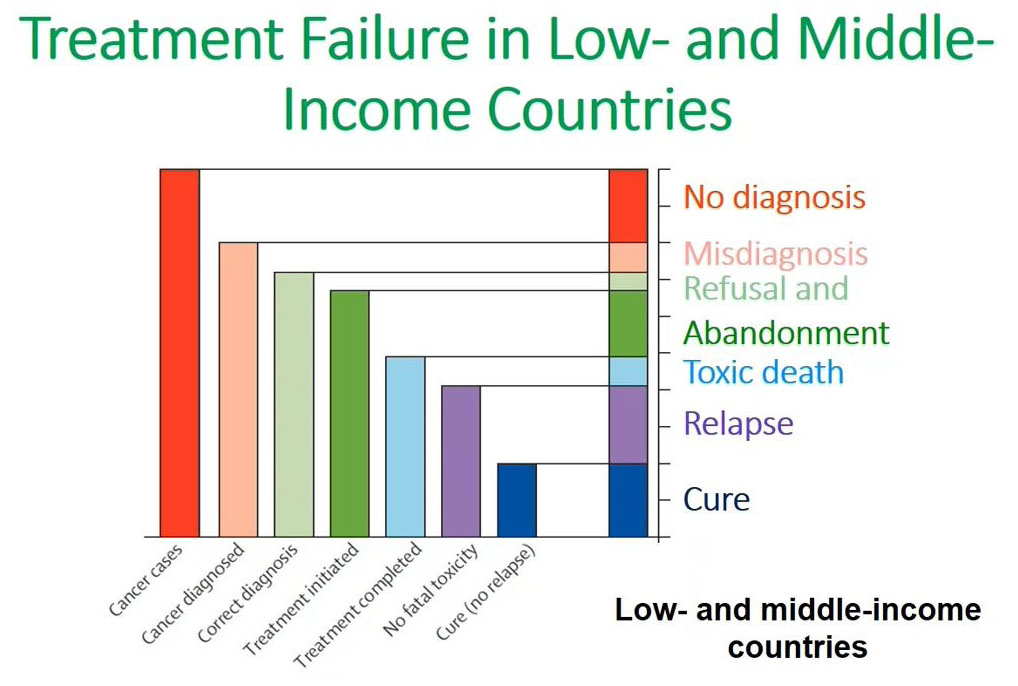
フローサイトメトリーによるイムノフェノタイピング(FCI)は、造血器腫瘍の臨床診断に大きな可能性を持ちながら、まだ十分には活用できていません。現在でも自家調製検査法(Laboratory Developed Test, LDT)が多く使用されていることから、検査室には下図に示すような多くの課題が残ったままとなっています。
私たちは、世界中の検査室が、正確な結果を迅速に得られるようサポートすることを使命としています。そして、疾患と闘う患者と向き合う医療従事者のみなさんを全力で支えます。
規制要件への対応
ワークフロー効率を改善する方法

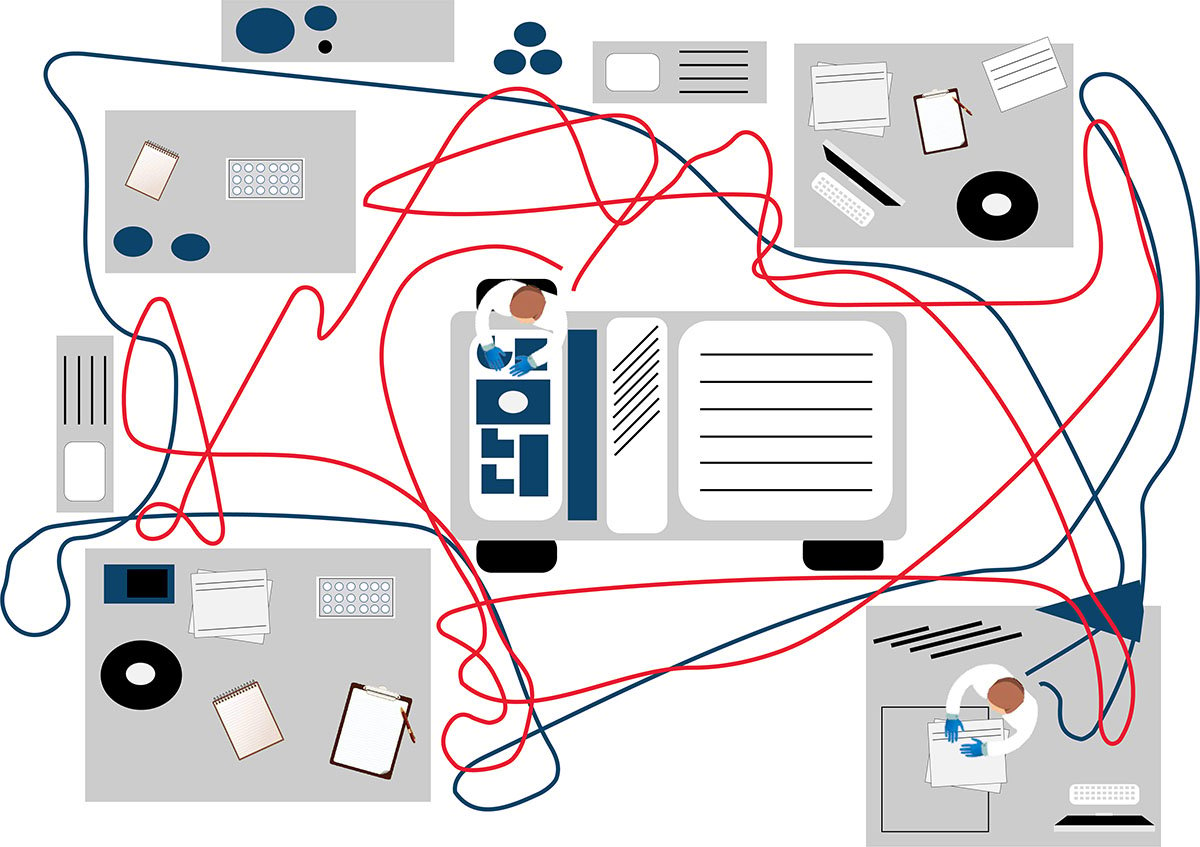
フローサイトメトリーによるイムノフェノタイピング(Flow cytometric immunophenotyping, FCI )の自家調製検査(LDT) では、パネルのスクリーニングに用いる抗体カクテルをマニュアルで作製します。マニュアル分注を伴うプロセスは、人的ミスや、ピペットのキャリブレーションが正しく行われていないことなどが原因でエラーが起こりやすいものです。また、液状試薬は、経時的に劣化するリスクを伴います。乾燥状態の試薬が事前調製(プレミクス)された標準化抗体パネルの場合、調製工程での上記のようなリスクが取り除かれます。コントロール細胞とあらかじめ調整された標準化検査パネルを使用することで、検査結果に確信を持つことが可能です。
フローサイトメーターの購入に伴うコストは高額であるため、この投資に対するリターンを最大限に高めるには、フローサイトメーターで実施する検査の効率と精度を確保しなければなりません。
自家調製検査(LTD)の抗体調製は非常に複雑なことから、フローサイトメトリーのパネル調製を専門に担う技師を配置する検査室もあり、コストが非常に高くなる一因となっています。一方、標準化された試薬では、マニュアルでの調製作業が不要で、ムダやエラーが減り、大きな節約につなげることが可能です。
より少ない労力・コストで、より多くの検査を実施するには? ClearLLabによるコスト削減についてはこちらをご覧ください!
自家調製検査(LDT)では、検査室独自で開発したパネルをマニュアルで作製して使用することが多く、その場合、それぞれの抗体を個々に調製した後、バリデーションします。これが終わればカクテルを作製し、抗体を組み合わせたカクテルとしての特異性を再バリデーションしなければなりません。抗体を混合したことでバリデーション結果に影響が出てしまった場合は、さらに調製が必要になることもあります。通常のバッチテスト、安定性テストに加え、品質保証全般で、各国の規制を遵守し、適正に文書を管理しなければなりません。調製にかかるこうした作業は、最先端の10カラーフローサイトメーターを使用している場合は特に、ワークフローの大きな部分を占めることになります。標準化された試薬の使用することで、ワークフローが短縮され、より早く結果が得られるだけでなく、手作業による繰り返し作業をなくし、データ解析に注力することが可能になります。
検査のセットアップや結果解析が複雑でばらつきがあることが、最近まで、臨床データの共有と教育の障壁となっていました。異なる施設で行われた検査の場合、同じコンセンサスガイドラインに基づいて行われたとしても、標準化された単一のアプローチが使用されていなければ、結果を施設間で比較することはできません。世界規模で標準化された試薬および手順を用いて検査を行うことで、共同研究、データ共有、教育の体制が整い、臨床教育と臨床研究の質が共に向上します。標準化されたアプローチを用いて得られた結果は、国際ガイドラインへの準拠が確保され、プロトコルのセットアップや結果の解釈のばらつき等の交絡要因が取り除かれます。
確信を持てる結果をあなたとあなたの患者さまに。的確な結果が得られる検査手法
臨床医が正しい診断、適切な治療決定を行うには、検査結果が正確である必要があります。検体検査結果の解釈や症例結論が適切でない場合、患者にとって深刻な結果が生じる可能性があります。標準化された検査では、臨床医は得られた結論について自信を深めることができ、その検査が仮に別の場所で行われたとしても、同じ結論が導かれたであろう、と認識できることで、患者に質の高い医療を提供することが可能です。患者にとっては、的確な検査結果をより速く得られることにより、適切な治療を可能な限り早期に開始することが可能となることから、良い転帰につながる可能性が高まります。
ClearLLab 10Cのワークフロー
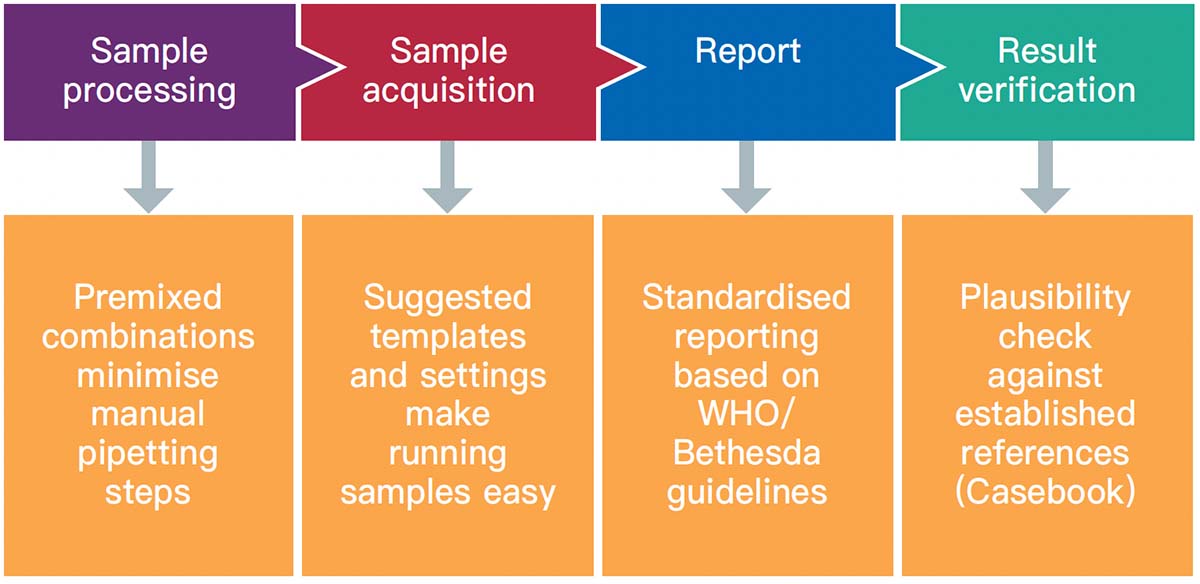
独自のテクノロジーであるDURAイノベーションテクノロジーを用いたClearLLab 10Cは事前調製済み(プレミクス)のドライ抗体試薬パネルであり、検査室での調製工程で起こり得るヒューマンエラーを減らすことが可能です。ClearLLab 10C を用いることにより、14の工程を削減、5以上の工程を簡略化できることから、結果を得るまでの時間が短縮されます。
ClearLLab 10Cのデザイン
ClearLLab 10Cの構成

*非ホジキンリンパ腫のみに対応
参考資料
- Bray F, Ferlaym J et al. (2018), Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 68: 394-424. doi:10.3322/caac.21492
- American Society of Hematology. Leukemia (n.d.). https://www.hematology.org/education/patients/blood-cancers/leukemia Accessed December 02,2021
- American Society of Hematology. Lymphoma (n.d.). https://www.hematology.org/education/patients/blood-cancers/lymphoma Accessed December 02,2021
- American Society of Hematology. Myeloma (n.d.). https://www.hematology.org/education/patients/blood-cancers/myeloma Accessed December 02,2021
- Arber DA, Orazi A et al. (2016), The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood, 127(20); 2391-2405. doi: 10.1182/blood-2016-03-643544 https://ashpublications.org/blood/article/127/20/2391/35255/The-2016-revision-to-the-World-Health-Organization
- Swerdlow SH, Campo E et al. (2016), The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood, 127(20): 2375-2390. doi: 10.1182/blood-2016-01-643569 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4874220/