IVD-Regulation (IVDR)

Like many other ideas that are intended to improve existing products or procedures, the new European IVD Regulation (IVD-R) 2017/746 road is paved with good intentions. However, some of these intentions may potentially render the daily life of clinical flow cytometry laboratories a lot more complex than what they are now, due to the latest regulations for Laboratory-Developed Tests (LDTs).
We at Beckman Coulter Life Sciences are partnering with you and support you with the largest available portfolio of CE-IVD and IVDR compliant reagents in the market and support you in your patients' journey in order to bring transparency into this important change.
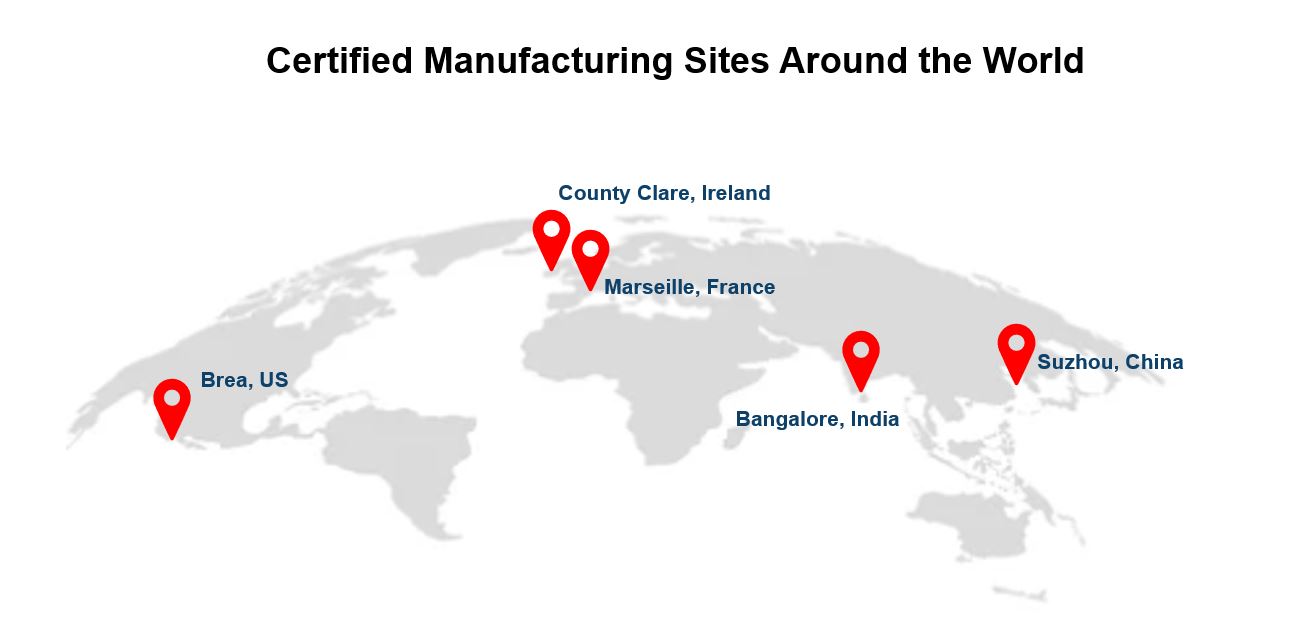
More than 250 associates are working diligently across the globe to ensure that our product portfolio is ready for the new IVDR. In 2023 we have grown our robust network to five certified manufacturing sites to keep up with the demand.
Find Your IVDR Products
Introduction to IVDR Basics
The In Vitro Diagnostic Regulation (IVDR), which went into effect in 2017, creates a robust and transparent framework of rules that improve clinical safety and reduces the risk of discrepancies in interpretations across the EU. It introduces stricter regulations for medical devices, lab tests, and assay kits, and for the first time, covering in-house laboratory-developed tests (LDTs). Manufacturers and laboratories were expected to be compliant by May 2022. But the COVID-19 pandemic created another challenge in the marathon toward compliance to meet that deadline. As a result, the EU gave an extension on the time limit for IVDR compliance.
Revised timeline for Laboratories to implement different aspects of IVDR
Resources to help your lab make a smooth transition to the IVDR
Unlike the previous IVD directive, the new regulation will include in-house IVD tests performed by the laboratories. This means that laboratories must follow standards that are, in many ways, similar to those of manufacturers. As a starting point, laboratories should audit all tests performed in the lab and ask questions such as:
Laboratories that choose to manufacture LDTs due to a lack of a viable alternative must develop a Quality Management System (QMS) that is compliant with the ISO-15189 “Medical laboratories — Requirements for quality and competence.” It requires the lab to get accredited for each single measurement/analysis they do. Monitoring and recording test performance, maintaining documentation about the test's manufacture and use, and justifying why omitted requirements are not applicable, are all part of the QMS.
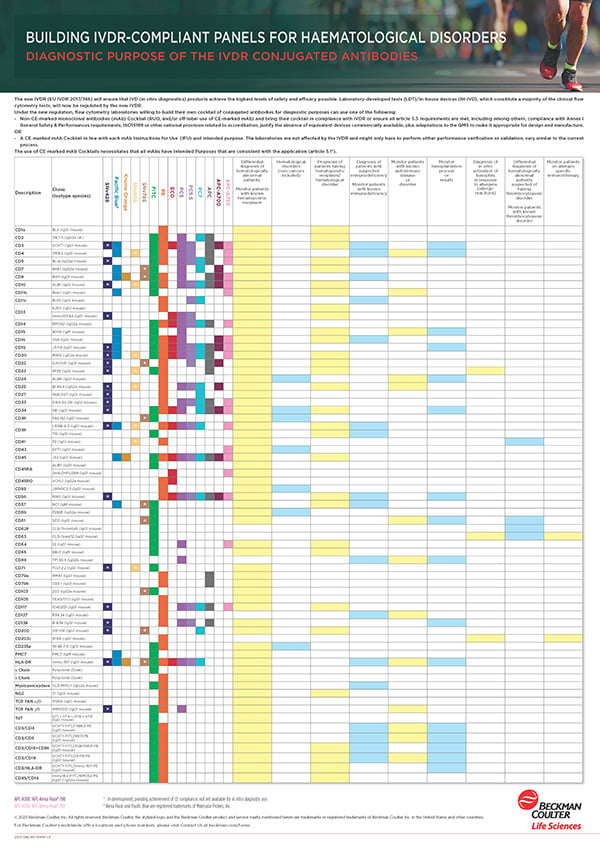
The following whitepapers are a starting point to set up your lab for IVDR-compliant creation of lab-developed tests using RUO products or CE-IVD products deviating from the IFU.